About Our Lab
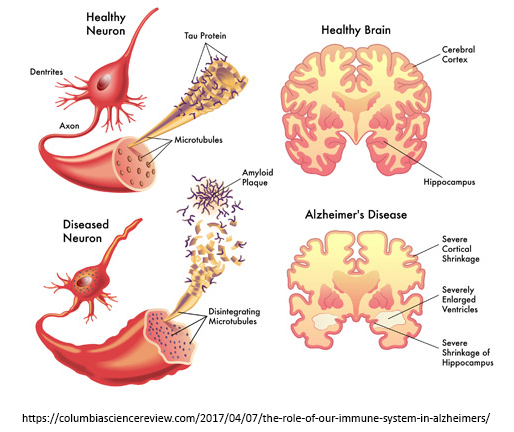
Our research focuses on the mechanisms of neurodegeneration in Alzheimer's disease (AD) and related neurological disorders such as Frontotemporal dementia (FTD) and Amyotrophic Lateral Sclerosis (ALS). AD is the leading cause of dementia and most prevalent neurodegenerative disease that currently afflicts 5.4 million individuals in the US and 40 million people worldwide. Due to the aging population, the US estimate is expected to rise to 13.2 million by 2050 if no effective treatment is found. In 2012, the cost of caring for AD and related dementia patients in the US stood at $200 billion/yr, and this figure is projected to rise to $1.1 trillion/yr by 2050 (2012 dollars)1. Therefore, AD is a tremendous cost to patients and families in both human and financial terms. Pathologically, brains afflicted with AD are riddled with two highly toxic proteins, namely amyloid-beta (Aβ; senile plaques) and tau (tangles), which are the underlying causes of neurodegeneration in AD2-4. While Aβ is the major driver of tau (tangle) pathology, Aβ without the tau protein is unable to produce neurodegeneration in brain5-7. The major pathological hallmark of FTD-ALS spectrum disorders is the accumulation of a protein called TDP-438,9. While this protein normally resides in the nucleus of neurons, TDP-43 aberrantly accumulates in the cytoplasm of diseased neurons10-12. Under these conditions, TDP-43 becomes extremely toxic to mitochondria13-15, the energy-producing powerhouses of neurons. Incidentally, TDP-43 pathology is not unique to FTD-ALS disorders but is also commonly found in AD and other dementias16. Likewise, tau pathology (tauopathy) is a major feature of a subclass of FTD and multiple other tauopathies. Our ongoing research efforts are aimed at deciphering the molecular signaling pathways underlying aberrant protein accumulation and neurotoxicity in neurodegenerative diseases and utilizing this knowledge to therapeutically target these toxic pathways. Summarized below are several highlights of our ongoing research.
Aβ production and neurotoxicity
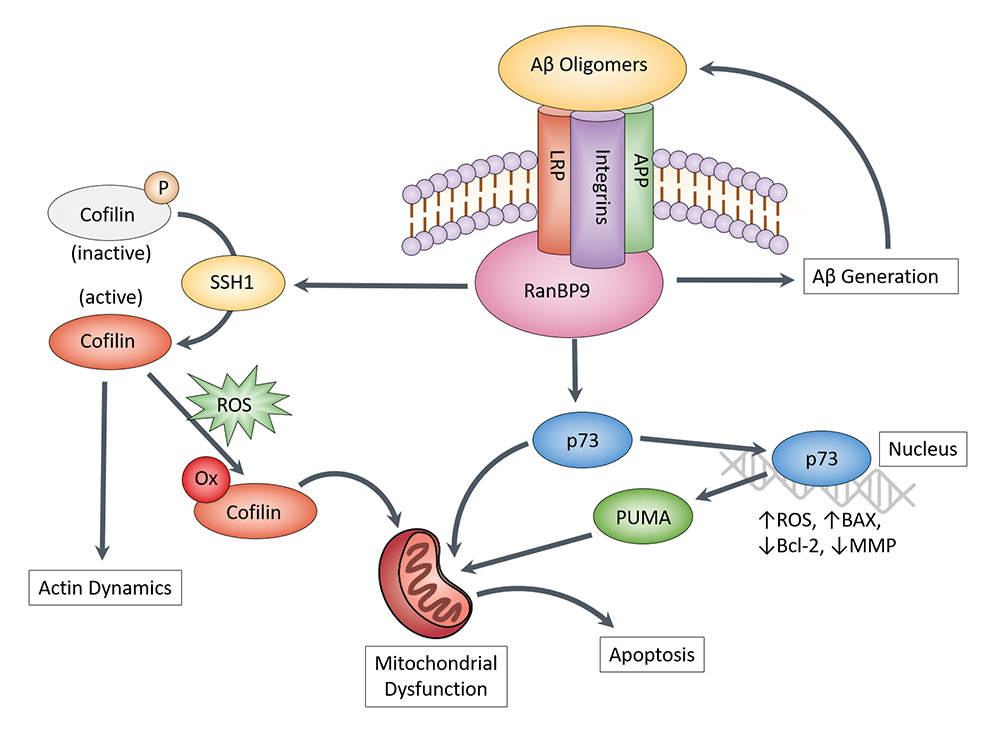
Aβ is small peptide derived from 2 proteolytic cuts (beta- & gamma-secretase) made from its precursor protein, APP. While Aβ is normally produced in small quantities, the abnormal accumulation of Aβ in AD (hence senile plaques) is determined by both the rate of production and clearance. While studying how Aβ is both produced and cleared via a receptor protein called LRP17-21, we identified a scaffolding protein RanBP9, which accounted for the effects of LRP in increasing Aβ production22,23. We later discovered that RanBP9 not only promotes Aβ production via the first cut (beta-secretase) of APP but is also necessary to mediate the toxic effects of Aβ on neurons24,25. This neurotoxic effect was found to require 2 others downstream proteins, namely cofilin25,26 and p7327. Both proteins exert their toxic effects directly on mitochondria, the energy-producing centers of neurons. We have also discovered that the enzyme SSH1, which activates cofilin and promotes cofilin translocation to mitochondria, is necessary for Aβ to produce its neurotoxic effects28,29. Hence, we are currently working on small molecule inhibitors of SSH1 (collaboration with Yu Chen & James Leahy), which we believe will be effective in blocking the neurotoxic actions of Aβ. We are also validating our studies in a mouse model of AD, utilizing genetically modified mice lacking SSH1.
Tau, chaperones, and neurodegeneration
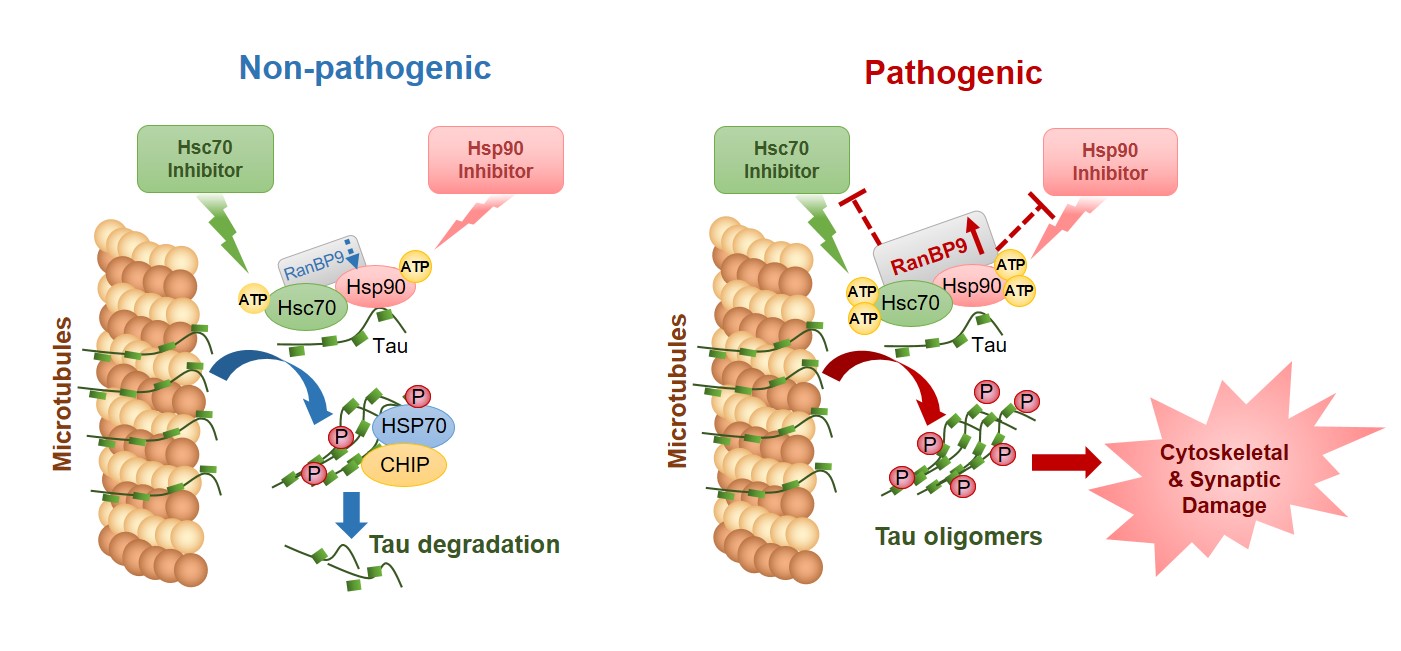
Tau is a microtubule-associated protein known to promote the assembly and stability of microtubules (MTs)30-32 . MTs function as a network of dynamic ‘railway’ system for neurons, on which many different forms of cargo are transported along the length and branches of neurons. Hence, the stability of such a ‘railway’ network is critical for a wide variety of neuronal functions. In AD and other tauopathies, tau falls off the MT tracks, fails to stabilize the MTs, and becomes prone to self-aggregation (i.e. tangles). Often tau undergoes multiple forms of modification, including phosphorylation (addition of phosphate group), and misfolded and aggregated tau usually contains excessive amounts of phosphorylation. Chaperone proteins, such as Hsp90 and Hsp70/Hsc70, normally function to detect misfolded proteins (i.e. tau). Once detected, these chaperone complexes can either attempt to refold/preserve tau or triage tau for degradation33-37 . In AD and other tauopathies, the chaperone system appears to engage tau for refolding but is unable to do so, ultimately leading to the preservation of misfolded tau. Our recent studies have discovered that RanBP9, which is highly elevated in brains of AD patients and AD animal models23,25,29, functions to preserve misfolded tau via forming a complex with the Hsp90 and Hsc70 chaperone system. Our findings indicate that this regulatory interplay may in part explain why the Hsp90/Hsc70 chaperone system is unable to disengage tau and route it to the degradation pathway. This has important therapeutic implications, as laboratories worldwide are currently pursuing the development of multiple Hsp90 and Hsc70 inhibitor compounds for therapeutic intervention. Our ongoing research efforts are aimed at understanding how the chaperone system engages with various components of the RanBP9 and SSH1 pathway to regulate triaging of misfolded tau.
TDP-43, CHCHD10, and mitochondrial dysfunction
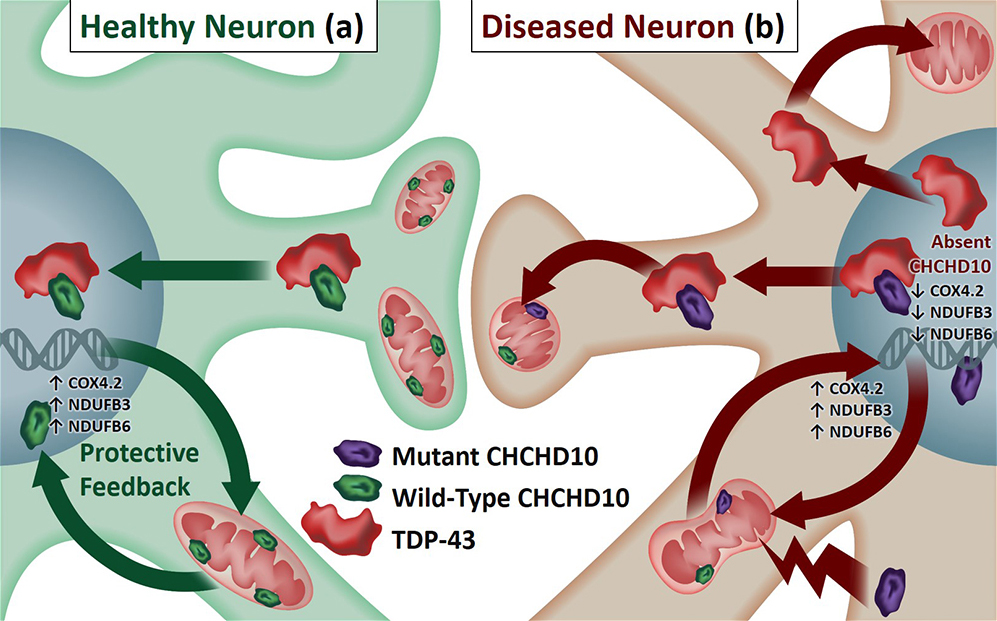
Accumulation of cytoplasmic TDP-43 represents a common pathological link between FTD and ALS8,9, and common genetic mutations in multiple genes are associated with both FTD and ALS38,39. One of these genes recently discovered is CHCHD10, a nuclear gene coding for a mitochondrial protein found in both ALS and FTD patients. However, neither the normal function of endogenous CHCHD10 nor its role in the pathological milieu (i.e. TDP-43 pathology) of FTD/ALS are understood. In our most recent work, we made a series of observations utilizing C. elegans models (worms), mammalian cell lines, primary neurons, and mouse brains, demonstrating that CHCHD10 normally exerts a protective role in mitochondrial and synaptic integrity as well as in the retention of nuclear TDP-43, whereas FTD/ALS-associated mutations (R15L & S59L) exhibit loss of function phenotypes in C. elegans genetic complementation assays and dominant negative activities in mammalian systems, resulting in mitochondrial/synaptic damage and cytoplasmic TDP-43 accumulation. As such, our results provide a pathological link between CHCHD10-associated mitochondrial/synaptic dysfunction and cytoplasmic TDP-43 inclusions. Our ongoing research efforts are aimed at understanding both the normal and pathological functions of CHCHD10 in the context of nuclear to mitochondria signaling as well as how TDP-43 and CHCHD10 coordinate their functions to regulate neuronal homeostasis, mitochondrial function, and longevity of the whole organism.
Autophagy, mitophagy, and accumulation of toxic components
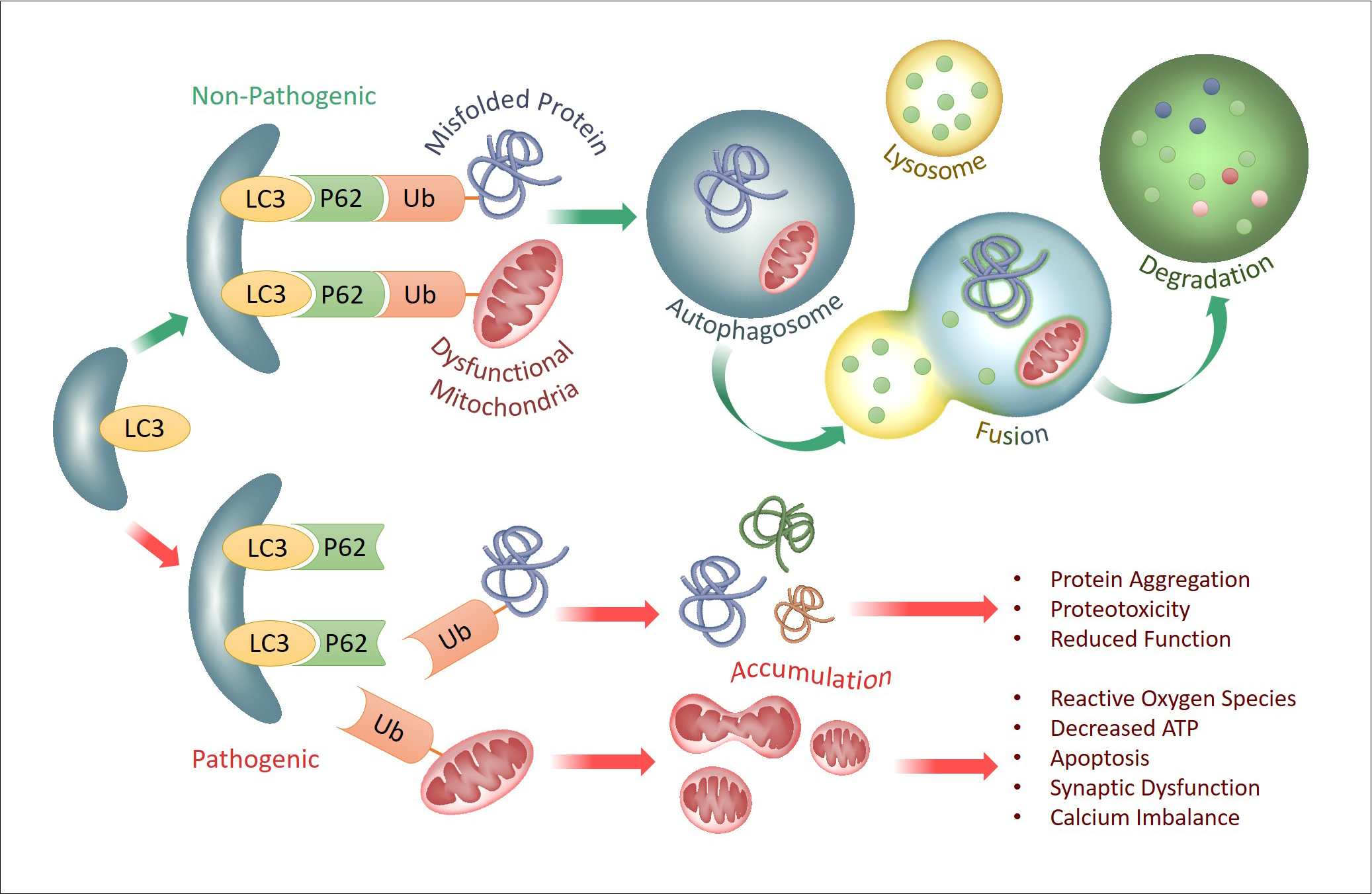
Autophagy is a natural and regulated destructive mechanism by which unnecessary or dysfunctional cell components (proteins or organelles) are targeted for degradation and recycling. Mitophagy is a form of selective autophagy that targets dysfunctional mitochondria for degradation; this process is vital for neuronal homeostasis and survival, as dysfunctional mitochondria are highly toxic to neurons and synapses. During selective autophagy, aberrantly misfolded proteins (i.e. tau or TDP-43) and/or dysfunctional mitochondria are recognized and targeted by a form of chemical modification called ubiquitination, and these ubiquitinated products are then recognized by autophagy cargo receptors (i.e. p62 and others) to be packaged into cellular vesicles called autophagosomes. Autophagosomes then fuse with lysosomes where the contents are degraded by protease enzymes. These degraded components can then be released and be reused by cells to rebuild cellular proteins and structures. While many short-lived proteins are rapidly destroyed by a highly efficient degradation machinery called the proteasome, more long-lived misfolded proteins (i.e. tau, TDP-43) and dysfunctional mitochondria are preferentially degraded via selective autophagy. Mounting evidence to date indicates that the accumulation of these toxic misfolded proteins in neurodegenerative diseases is at least in part due to ineffective or overwhelmed autophagy machinery. This defect can come at multiple levels of the autophagy, including ubiquitination, recognition of ubiquitinated products, formation of autophagosomes, fusion of autophagosomes to lysosomes, to lysosomes themselves. Our ongoing research aims to better understand how various misfolded proteins (tau, Aβ, TDP-43, & others) and dysfunctional mitochondria are handled through the autophagy pathway, in the hopes of enhancing the autophagy machinery through specific molecular steps in the process.
Extracellular vesicles as disease biomarkers
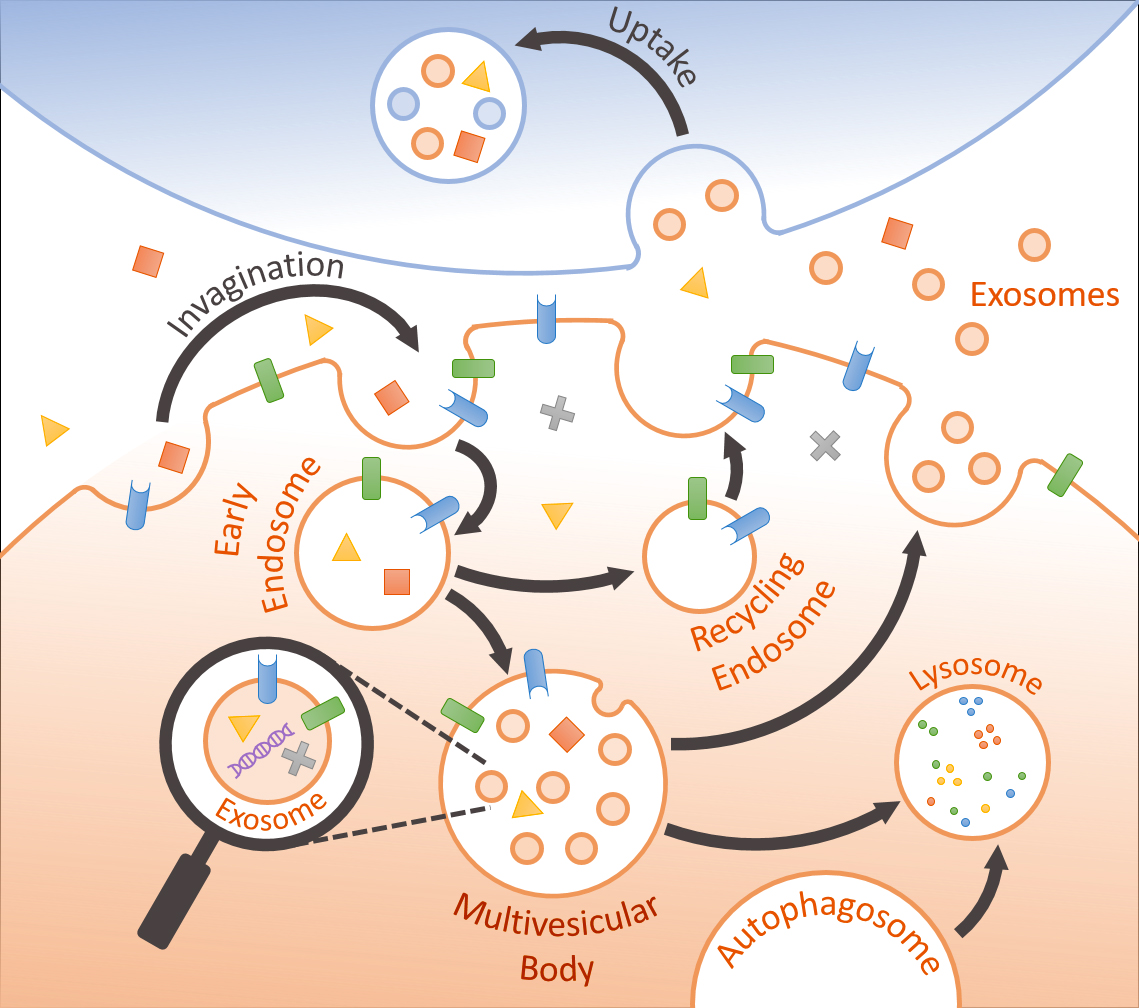
Emerging evidence indicates that release and uptake of extracellular vesicles (EVs) represents an important form of intercellular communication that could transmit beneficial or pathogenic signals across different cells40. For example, normal EVs have been shown to regulate neuronal neurite outgrowth, survival, and synaptic activity41,42 as well as microglial activation43,44. EVs are secreted by all cell types and contain specific sets of RNA and proteins (membrane & cytosolic)45,46. Large microvesicles (MVs) (150-1000nm) are derived from evagination and excision of the plasma membrane, whereas the smaller exosomes (30-150nm) are intraluminal vesicles (ILVs) formed by inward budding of the limiting membrane of late endosomes or multivesicular bodies (MVBs), and are secreted by fusion with the plasma membrane47. Despite their ubiquitous presence in brain, the role of EVs in brain cells (i.e. neurons & glia) and their potential effects on neurodegenerative processes have largely been unexplored. In pathological settings, proteins prone to aggregation (i.e. Ab, tau, a-synuclein, & prion) are released in EVs, potentially providing an avenue for the spread of such misfolded proteins to neighboring healthy cells48-51. The working hypothesis of our research is that the content of brain-derived EVs, particularly exosomes, recapitulate the overall physiological or pathological milieu of the brain captured in a micro-environment. As exosomes are capable of crossing the blood-brain-barrier, accurate assessment of brain-derived exosomes in blood plasma may serve as effective and noninvasive biomarkers of brain disease. For this purpose, our research is aimed at profiling and categorizing the protein content (proteome) of EVs in normal versus pathological conditions. In addition, we are assessing the neurotoxic or neuroprotective properties of such EVs.

REFERENCES
- 2013 Alzheimer's Disease Facts and Figures. Alzheimer's Association.
- Hardy, J. A. & Higgins, G. A. Alzheimer's disease: the amyloid cascade hypothesis. Science 256, 184-185 (1992).
- Hardy, J. & Selkoe, D. J. The amyloid hypothesis of Alzheimer's disease: progress and problems on the road to therapeutics. Science 297, 353-356, doi:10.1126/science.1072994 (2002).
- Vassar, R., Kovacs, D. M., Yan, R. & Wong, P. C. The beta-secretase enzyme BACE in health and Alzheimer's disease: regulation, cell biology, function, and therapeutic potential. The Journal of neuroscience : the official journal of the Society for Neuroscience 29 12787-12794, doi:10.1523/JNEUROSCI.3657-09.2009 (2009).
- Jin, M. et al. Soluble amyloid beta-protein dimers isolated from Alzheimer cortex directly induce Tau hyperphosphorylation and neuritic degeneration. Proceedings of the National Academy of Sciences of the United States of America 108, 5819-5824, doi:10.1073/pnas.1017033108 (2011).
- Rapoport, M., Dawson, H. N., Binder, L. I., Vitek, M. P. & Ferreira, A. Tau is essential to beta -amyloid-induced neurotoxicity. Proceedings of the National Academy of Sciences of the United States of America 99, 6364-6369, doi:10.1073/pnas.092136199 (2002).
- Vossel, K. A. et al. Tau reduction prevents Aβ-induced defects in axonal transport. Science 330, 198, doi:10.1126/science.1194653 (2010).
- Weishaupt, J. H., Hyman, T. & Dikic, I. Common Molecular Pathways in Amyotrophic Lateral Sclerosis and Frontotemporal Dementia. Trends in molecular medicine 22, 769-783, doi:10.1016/j.molmed.2016.07.005 (2016).
- Janssens, J. & Van Broeckhoven, C. Pathological mechanisms underlying TDP-43 driven neurodegeneration in FTLD-ALS spectrum disorders. Human molecular genetics 22, R77-87, doi:10.1093/hmg/ddt349 (2013).
- Igaz, L. M. et al. Expression of TDP-43 C-terminal Fragments in Vitro Recapitulates Pathological Features of TDP-43 Proteinopathies. The Journal of biological chemistry 284, 8516-8524, doi:10.1074/jbc.M809462200 (2009).
- Yang, C. et al. The C-terminal TDP-43 fragments have a high aggregation propensity and harm neurons by a dominant-negative mechanism. PloS one 5, e15878, doi:10.1371/journal.pone.0015878 (2010).
- Zhang, Y. J. et al. Aberrant cleavage of TDP-43 enhances aggregation and cellular toxicity. Proceedings of the National Academy of Sciences of the United States of America 106, 7607-7612, doi:10.1073/pnas.0900688106 (2009).
- Wang, W. et al. The ALS disease-associated mutant TDP-43 impairs mitochondrial dynamics and function in motor neurons. Human molecular genetics 22, 4706-4719, doi:10.1093/hmg/ddt319 (2013).
- Wang, W. et al. The inhibition of TDP-43 mitochondrial localization blocks its neuronal toxicity. Nature medicine 22, 869-878, doi:10.1038/nm.4130 (2016).
- Xu, Y. F. et al. Wild-type human TDP-43 expression causes TDP-43 phosphorylation, mitochondrial aggregation, motor deficits, and early mortality in transgenic mice. The Journal of neuroscience : the official journal of the Society for Neuroscience 30, 10851-10859, doi:10.1523/JNEUROSCI.1630-10.2010 (2010).
- Josephs, K. A. et al. TAR DNA-binding protein 43 and pathological subtype of Alzheimer's disease impact clinical features. Annals of neurology 78, 697-709, doi:10.1002/ana.24493 (2015).
- Yoon, I. S. et al. Low-density lipoprotein receptor-related protein promotes amyloid precursor protein trafficking to lipid rafts in the endocytic pathway. FASEB journal : official publication of the Federation of American Societies for Experimental Biology 21, 2742-2752, doi:10.1096/fj.07-8114com (2007).
- Kang, D. E. et al. Genetic association of the low-density lipoprotein receptor-related protein gene (LRP), an apolipoprotein E receptor, with late-onset Alzheimer's disease. Neurology 49, 56-61 (1997).
- Kang, D. E. et al. Modulation of amyloid beta-protein clearance and Alzheimer's disease susceptibility by the LDL receptor-related protein pathway. The Journal of clinical investigation 106, 1159-1166, doi:10.1172/JCI11013 (2000).
- Lakshmana, M. K., Chen, E., Yoon, I. S. & Kang, D. E. C-terminal 37 residues of LRP promote the amyloidogenic processing of APP independent of FE65. Journal of cellular and molecular medicine 12, 2665-2674, doi:10.1111/j.1582-4934.2008.00320.x (2008).
- Yoon, I. S., Pietrzik, C. U., Kang, D. E. & Koo, E. H. Sequences from the low density lipoprotein receptor-related protein (LRP) cytoplasmic domain enhance amyloid beta protein production via the beta-secretase pathway without altering amyloid precursor protein/LRP nuclear signaling. The Journal of biological chemistry 280, 20140-20147, doi:10.1074/jbc.M413729200 (2005).
- Lakshmana, M. K. et al. Novel role of RanBP9 in BACE1 processing of amyloid precursor protein and amyloid beta peptide generation. J.Biol.Chem. 284, 11863-11872, doi:M807345200 [pii];10.1074/jbc.M807345200 [doi] (2009).
- Lakshmana, M. K. et al. A fragment of the scaffolding protein RanBP9 is increased in Alzheimer's disease brains and strongly potentiates amyloid-beta peptide generation. FASEB J. 24, 119-127, doi:fj.09-136457 [pii];10.1096/fj.09-136457 [doi] (2010).
- Woo, J. A., Roh, S. E., Lakshmana, M. K. & Kang, D. E. Pivotal role of RanBP9 in integrin-dependent focal adhesion signaling and assembly. FASEB J. 26, 1672-1681, doi:fj.11-194423 [pii];10.1096/fj.11-194423 [doi] (2012).
- Woo, J. A. et al. Pivotal role of the RanBP9-cofilin pathway in Aβ-induced apoptosis and neurodegeneration. Cell death and differentiation 19, 1413-1423, doi:10.1038/cdd.2012.14 (2012).
- Roh, S. E. et al. Mitochondrial dysfunction and calcium deregulation by the RanBP9-cofilin pathway. FASEB journal : official publication of the Federation of American Societies for Experimental Biology 27, 4776-4789, doi:10.1096/fj.13-234765 (2013).
- Liu, T., Roh, S. E., Woo, J. A., Ryu, H. & Kang, D. E. Cooperative role of RanBP9 and P73 in mitochondria-mediated apoptosis. Cell death & disease 4, e476, doi:10.1038/cddis.2012.203 (2013).
- Woo, J. A. et al. Slingshot-Cofilin activation mediates mitochondrial and synaptic dysfunction via Aβ ligation to beta1-integrin conformers. Cell death and differentiation 22, 921-934, doi:10.1038/cdd.2015.5 (2015).
- Woo, J. A. et al. RanBP9 at the intersection between cofilin and Aβ pathologies: rescue of neurodegenerative changes by RanBP9 reduction. Cell death & disease 6, 1676, doi:10.1038/cddis.2015.37 (2015).
- Mandelkow, E. M. et al. Tau domains, phosphorylation, and interactions with microtubules. Neurobiology of aging 16, 355-362; discussion 362-353 (1995).
- Mandelkow, E. M. & Mandelkow, E. Biochemistry and cell biology of tau protein in neurofibrillary degeneration. Cold Spring Harbor perspectives in medicine 2, a006247, doi:10.1101/cshperspect.a006247 (2012).
- Takalo, M., Salminen, A., Soininen, H., Hiltunen, M. & Haapasalo, A. Protein aggregation and degradation mechanisms in neurodegenerative diseases. American journal of neurodegenerative disease 2, 1-14 (2013).
- Dou, F. et al. Chaperones increase association of tau protein with microtubules. Proceedings of the National Academy of Sciences of the United States of America 100, 721-726, doi:10.1073/pnas.242720499 (2003).
- Blair, L. J. et al. Accelerated neurodegeneration through chaperone-mediated oligomerization of tau. The Journal of clinical investigation 123, 4158-4169, doi:10.1172/JCI69003 (2013).
- Dickey, C. A. et al. The high-affinity HSP90-CHIP complex recognizes and selectively degrades phosphorylated tau client proteins. The Journal of clinical investigation 117, 648-658, doi:10.1172/JCI29715 (2007).
- Jinwal, U. K. et al. Hsc70 rapidly engages tau after microtubule destabilization. The Journal of biological chemistry 285 16798-16805, doi:10.1074/jbc.M110.113753 (2010).
- Fontaine, S. N. et al. The active Hsc70/tau complex can be exploited to enhance tau turnover without damaging microtubule dynamics. Human molecular genetics 24, 3971-3981, doi:10.1093/hmg/ddv135 (2015).
- Burrell, J. R. et al. The frontotemporal dementia-motor neuron disease continuum. Lancet 388, 919-931, doi:10.1016/S0140-6736(16)00737-6 (2016).
- Ng, A. S., Rademakers, R. & Miller, B. L. Frontotemporal dementia: a bridge between dementia and neuromuscular disease. Annals of the New York Academy of Sciences 1338, 71-93, doi:10.1111/nyas.12638 (2015).
- Rajendran, L. et al. Emerging roles of extracellular vesicles in the nervous system. The Journal of neuroscience : the official journal of the Society for Neuroscience 34, 15482-15489, doi:10.1523/JNEUROSCI.3258-14.2014 (2014).
- Wang, S. et al. Synapsin I is an oligomannose-carrying glycoprotein, acts as an oligomannose-binding lectin, and promotes neurite outgrowth and neuronal survival when released via glia-derived exosomes. The Journal of neuroscience : the official journal of the Society for Neuroscience 31, 7275-7290, doi:10.1523/JNEUROSCI.6476-10.2011 (2011).
- Antonucci, F. et al. Microvesicles released from microglia stimulate synaptic activity via enhanced sphingolipid metabolism. The EMBO journal 31, 1231-1240, doi:10.1038/emboj.2011.489 (2012).
- Prada, I., Furlan, R., Matteoli, M. & Verderio, C. Classical and unconventional pathways of vesicular release in microglia. Glia 61, 1003-1017, doi:10.1002/glia.22497 (2013).
- Verderio, C. et al. Myeloid microvesicles are a marker and therapeutic target for neuroinflammation. Annals of neurology 72, 610-624, doi:10.1002/ana.23627 (2012).
- Valadi, H. et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nature cell biology 9, 654-659, doi:10.1038/ncb1596 (2007).
- Skog, J. et al. Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nature cell biology 10, 1470-1476, doi:10.1038/ncb1800 (2008).
- Raposo, G. & Stoorvogel, W. Extracellular vesicles: exosomes, microvesicles, and friends. The Journal of cell biology 200, 373-383, doi:10.1083/jcb.201211138 (2013).
- Rajendran, L. et al. Alzheimer's disease beta-amyloid peptides are released in association with exosomes. Proceedings of the National Academy of Sciences of the United States of America 103, 11172-11177, doi:10.1073/pnas.0603838103 (2006).
- Danzer, K. M. et al. Exosomal cell-to-cell transmission of alpha synuclein oligomers. Molecular neurodegeneration 7, 42, doi:10.1186/1750-1326-7-42 (2012).
- Saman, S. et al. Exosome-associated tau is secreted in tauopathy models and is selectively phosphorylated in cerebrospinal fluid in early Alzheimer disease. The Journal of biological chemistry 287, 3842-3849, doi:10.1074/jbc.M111.277061 (2012).
- Vella, L. J. et al. Packaging of prions into exosomes is associated with a novel pathway of PrP processing. The Journal of pathology 211, 582-590, doi:10.1002/path.2145 (2007).




